Abstract
Background: Mutational (RAS, PTPN11 etc.) or non-mutational activation of RAS/RAF/ERK pathway is associated with primary or secondary resistance to venetoclax in AML therapy (Zhang et al., Sig Transduct Target Ther. 2022). While upregulation of alternate antiapoptotic proteins e.g. Mcl-1 has been implicated in resistance to venetoclax (Zhang et al., Sig Transduct Target Ther. 2022) role of mitochondrial dynamics (fission/fusion) in venetoclax resistance has not been well explored. Oncogenic RAS activates downstream ERK1/2 phosphorylation and mitochondrial fission through ERK1/2-mediated phosphorylation of the dynamin-like mitochondrial fission GTPase Drp1 (Nagdas et al., Cell Rep. 2019). Drp1 deletion inhibits tumorigenesis of KRAS driven pancreatic cancer, but the importance of RAS driven changes in mitochondrial dynamics in AML is poorly defined. ERK1/2 inhibition may therefore provide insights into the dependence of AML cells on altered mitochondrial dynamics and metabolism driven by oncogenic RAS.
Objectives: We assessed the synergistic anti-leukemia efficacy of concomitant Bcl2 and ERK1/2 inhibition in preclinical models of venetoclax resistance and in primary patient samples (n=8). Further, we investigated the role of ERK1/2/Drp1-dependent mitochondrial fission in mediating resistance to venetoclax through altered metabolism.
Results: ERK1/2 inhibition using Compound 27 (Heightman et al., J Med Chem. 2018) a close analog of ASTX029, which is currently under clinical investigation in solid tumors (NCT03520075), was highly synergistic with venetoclax at inducing apoptosis in AML cells. NRAS-mutant OCI-AML3 and THP1 cells, that are intrinsically resistant to venetoclax, were sensitized to venetoclax by ERK1/2 inhibition with a combination index (CI) of 0.008 (cell death ~90% for combination versus 20% with venetoclax alone). Synergy was also confirmed in OCI-AML2 cells with acquired resistance to venetoclax, resulting in 75% apoptosis versus 22% by venetoclax alone (CI=0.6). The combination was also effective in eliminating bulk and CD34+CD38- leukemia initiating cells (LICs) in primary AML samples (CI:0.03-0.23). Colony formation assay and single cell level CyTOF analysis using an NRAS-mutant PDX showed markedly impaired clonogenic potential (p=0.0002) and decreased expression of cMyc and CD44 in LICs in response to single agent ERK1/2 inhibition and combination treatment, indicating a potential impact on stemness.
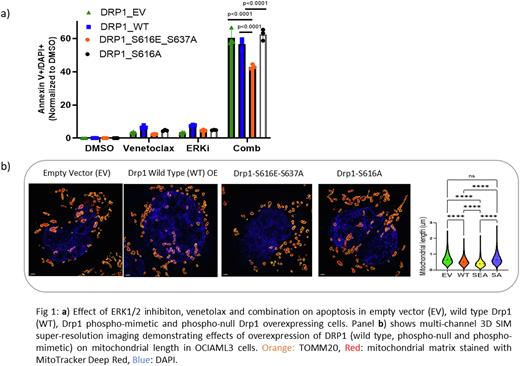
Mitochondrial reactive oxygen species (ROS) mitigation, Drp1-mediated mitochondrial fission and glycolysis are critical to maintain stemness in AML (Schimmer et al., Cell Stem Cell. 2018). As a result of ERK1/2 inhibition, pDrp1 (Ser616) levels decreased, and mitochondrial length (p<0.001) and mitochondrial ROS levels (p<0.01) were increased, suggesting impaired fission. In OCI-AML3 cells, combination treatment caused downregulation of Drp1 mRNA and decreased phosphorylation of Drp1 at Ser616. Rescue with overexpression of active phospho-mimetic form of Drp1 i.e. Drp1-Ser616Glu-Ser637Ala in OCI-AML3 cells resulted in decreased apoptosis and reversed the synergy between ERK1/2 and Bcl-2 inhibition when compared to wild type and phospho-null (Ser616Ala) Drp1 overexpression (p<0.001, Fig 1a). Of note, overexpression of Drp1-Ser616Glu-Ser637Ala phospho-mimetic led to decreased mitochondrial length (Fig 1b), suggesting enhanced fission with a distinct metabolic phenotype including reduced basal respiration, decreased maximal oxygen consumption rate (p<0.05) and ATP production (p<0.05), as well as reduced ROS production (p<0.01). Moreover, combination treatment failed to significantly induce ROS production in cells expressing the phospho-mimetic Drp1 as opposed to those expressing phospho-null or wildtype Drp1. Collectively, these results indicate that inhibition of ERK1/2/Drp1dependent mitochondrial fission and metabolic alterations may be a potential mechanism of synergistic tumor cell killing for the combination treatment.
Conclusion: ERK1/2 inhibition alters Drp1 dependent mitochondrial dynamics and metabolism, resulting in mitochondrial defects and overcoming resistance to venetoclax by causing apoptosis in venetoclax resistant AML cells. These data provide a strong rationale for the combination of ERK1/2 and Bcl-2 inhibitors in treatment of AML and warrant further investigation in the clinic.
Disclosures
Hindley:Astex Pharmaceuticals: Current Employment. Dao:Astex Pharmaceuticals, Inc.: Current Employment. Sims:Astex Pharmaceuticals: Current Employment. Andreeff:Chimerix: Current holder of stock options in a privately-held company; Oncolyze: Current holder of stock options in a privately-held company; German Research Council: Membership on an entity's Board of Directors or advisory committees; NCI: Membership on an entity's Board of Directors or advisory committees; Leukemia & Lymphoma Society: Membership on an entity's Board of Directors or advisory committees; Cancer UK: Membership on an entity's Board of Directors or advisory committees; Medicxi: Consultancy; Glycomimetics: Consultancy; Aptose: Consultancy, Membership on an entity's Board of Directors or advisory committees; Senti Bio: Consultancy, Research Funding; Syndax: Consultancy, Research Funding; Brooklyn ITX: Research Funding; Pinot Bio: Research Funding; Oxford Biomedical UK: Research Funding; AstraZeneca: Research Funding; Breast Cancer Research Foundation: Research Funding; Daiichi-Sankyo Inc.: Consultancy, Research Funding; Kintor Pharmaceutical: Research Funding; CLL Foundation: Membership on an entity's Board of Directors or advisory committees; Reata: Current holder of stock options in a privately-held company. Borthakur:Catamaran Bio, Abbvie, PPD Development, Protagonist Therapeutics, Janssen: Consultancy; Pacylex, Novartis, Cytomx, Bio Ascend: Membership on an entity's Board of Directors or advisory committees; Astex Pharmaceuticals, Ryvu, PTC Therapeutics: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal